Table of Contents
ToggleWhy we use Liquid Metal Batteries(LMB)?
To achieve carbon neutrality by 2060, increasing the use of non-fossil energy sources and expanding the use of renewable green technologies is essential. This includes establishing a circular economy and ensuring the widespread adoption of smart grid architecture.
Transitioning from fossil-fuel-powered plants to renewable sources, such as solar, hydro, and wind power, has the potential to significantly reduce carbon emissions in key industrial sectors. However, the intermittent nature and uneven geographical distribution of these renewable sources limit their widespread market dominance.
Thus, to mitigate grid inflexibility and maintain a stable energy supply, using robust energy storage systems is crucial. Grid-scale energy storage requires batteries with a prolonged operational lifetime of 20 to 30 years, exceptional safety, and low cost. Traditional batteries, such as Li-ion, Na-ion, lead-acid, and Zn-ion, experience capacity reduction because they employ solid electrodes and acidic or alkaline electrolytes.
This leads to phase transformation, dendrite growth, and side reactions, which pose challenges in meeting the durability standards required for grid-level energy storage. These limitations necessitate the exploration of alternative technologies to provide more reliable and sustainable energy storage solutions.
Liquid metal batteries (LMBs) present a novel approach to grid-level energy storage by utilizing liquid metals as both positive and negative electrodes. Metals such as antimony (Sb), bismuth (Bi), tin (Sn), lead (Pb), and tellurium (Te) are used for positive electrodes, while lithium (Li), sodium (Na), potassium (K), calcium (Ca), and magnesium (Mg) function as negative electrodes.
Principle of Liquid Metal Batteries(LMB)
Different from the traditional rechargeable batteries, liquid metal batteries (LMBs), also known as molten salt batteries, employ liquid metal for both their anodes and their cathodes, separated by a molten salt electrolyte, requiring no separators . LMBs are usually secondary batteries, and the structure of the battery is typically vertical, with the cathode, electrolyte, and anode from bottom to top.
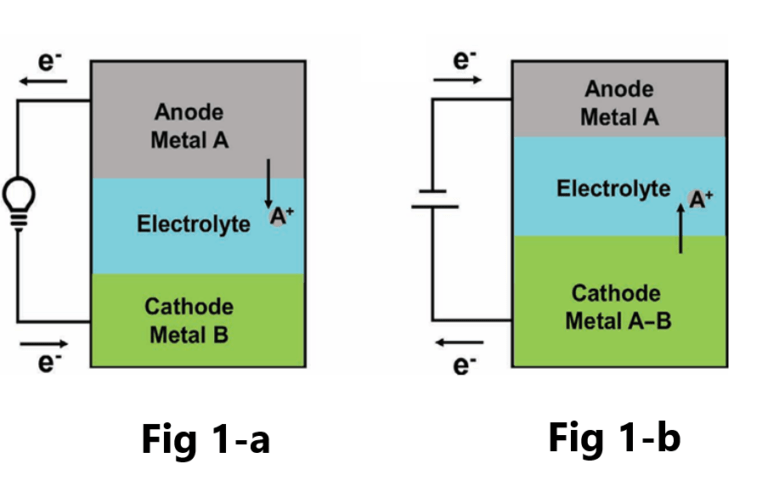
Normally, the density of the cathode is greater than that of the electrolyte, and the density of the electrolyte is greater than that of the anode . Also, these three materials should be immiscible, so the battery naturally forms a three-layer structure. The battery configuration is shown in Figure-1, where A and B rep resent the anode and cathode, respectively. During discharge, the anode metal is oxidized, and the resulting metal ions travel through the electrolyte to the cathode, forming an alloy at the cathode (Figure 1-a). While charging, A is reduced from the A-B alloy and migrates back to the anode (Figure 1-b). The whole process can be expressed as follows:
Discharging:
Anode- A—ze− → Az+;
Cathode B + Az+ + ze− → AB
Charging:
Anode Az+ + ze− → A
Cathode.; AB → B + Az+ + ze−.
At present, the most common anode and cathode are Li and Pb-Sb, respectively, with a stability of ~10 years and a Coulombic efficiency of ~73% . This combination is paired with an electrolyte of eutectic LiF-LiCl-LiI or LiCl-KCl. There are also some studies focusing on other metal systems to pursue higher energy density, lower cost, and lower operating temperatures . For example, Mg||Sb battery can reach a capacity of more than 1000 mAh/cm2 at 700°C, with a cost of 375 $kW/h (Li-based LMBs are lower than 100 $kW/h) . Besides, Ga has a melting point as low as 29.87°C and can form low-melting-point alloys with other metals. Thus, Wang et al. developed LMBs using Ga-Sn alloy, which has a melting point of 21°C. The resultant battery could be cycled at 35°C with a capacity of 409 mAh/g.
Features of LMBs
High-Rate Capability
Unlike solid-electrode batteries, which struggle with rapid charging, LMBs are capable of fast charging and discharging. Since the electrodes are in the liquid state, charge carrier diffusion is faster than that of solid-state electrodes . Additionally, the absence of a solid-liquid interface barrier between electrodes and electrolytes enables faster ion mobility. Moreover, the higher temperature also increases the reaction kinetics at the electrodes . The liquid metal layers allow for a uniform current distribution across the electrodes, preventing localized hotspots and ensuring the battery can handle high current loads without performance loss. Some studies indicate that fast charging has less impact on the battery than fast discharging because intermetallic compounds, which are usually accompanied by large volume changes, are formed on cathodes during discharging, while charging is not.
Long Lifetime
This is the most attractive advantage of LMBs. The liquid nature of the electrodes and electrolytes helps in maintaining consistent contact and reduces the formation of the dendrite and passivation layer . This self-healing property ensures the battery maintains high performance even under fast cycling conditions. Additionally, as there is no solid structure to crack or degrade, the battery’s lifespan is significantly extended. The stability of the electrolyte also contributes to the overall longevity of the batteries. The liquid electrolyte in these batteries is typically made from stable, high-temperature molten salts, which are less prone to decomposition or side reactions compared to the liquid electrolytes used in other battery types, such as lithium-ion batteries.
Low Cost
Most candidates for electrode materials are relatively common metals, so the price of the entire battery can be very low.
Scalability
Being able to respond rapidly to changes in electricity demand, LMBs are applied to grid-scale energy storage (0.1–1.0 GWh). They can also assist in frequency regulation, voltage support, and other grid services.
Disadvantages of LMBs
High Operating Temperature
LMBs typically operate at temperatures higher than 240°C. Maintaining stability in such a high-temperature environment is critical for safe operation. Otherwise, the overall efficiency of the system will be reduced. Also, since the materials are all liquid and the whole battery construction is based on their densities, the battery can not be moved freely.
Material Constraints
The requirements of melting points, density, and immiscibility limit the choice of electrodes and electrolytes. Also, the high operating temperature limits the materials for containment and insulation, as they must withstand extreme heat without degrading or reacting with the battery components.
Low Voltage and Low Energy Density
Due to the limitation of material combinations, the voltage of LMBs is relatively low. Typically, the voltage of LMBs is between 0.5 and 2.0 V (i.e., the Mg||Sb system is ~ 1.2 to 1.5 V, the Na||Pb system is ~ 0.9 to 1.2 volts, the Li||Pb-Sb is ~ 1–1.2 V, etc.). Compared to the lithium-ion battery, which produces a voltage of ~3.2–4.2 V, the volt age produced by LMBs is not satisfactory. The low voltage will result in low energy density.
Safety Concerns
The use of molten metals and salts introduces potential safety risks, particularly in breaking or corrosion in the containment system. A leak could result in high-temperature molten materials escaping, which could be hazardous.